60Co gamma ray induced mutants of cowpea and assessment of genetic variability by SCoT marker
DOI:
https://doi.org/10.14719/pst.1623Keywords:
Cowpea, Mutant, SCoT Marker, Genetic variation, PolymorphismAbstract
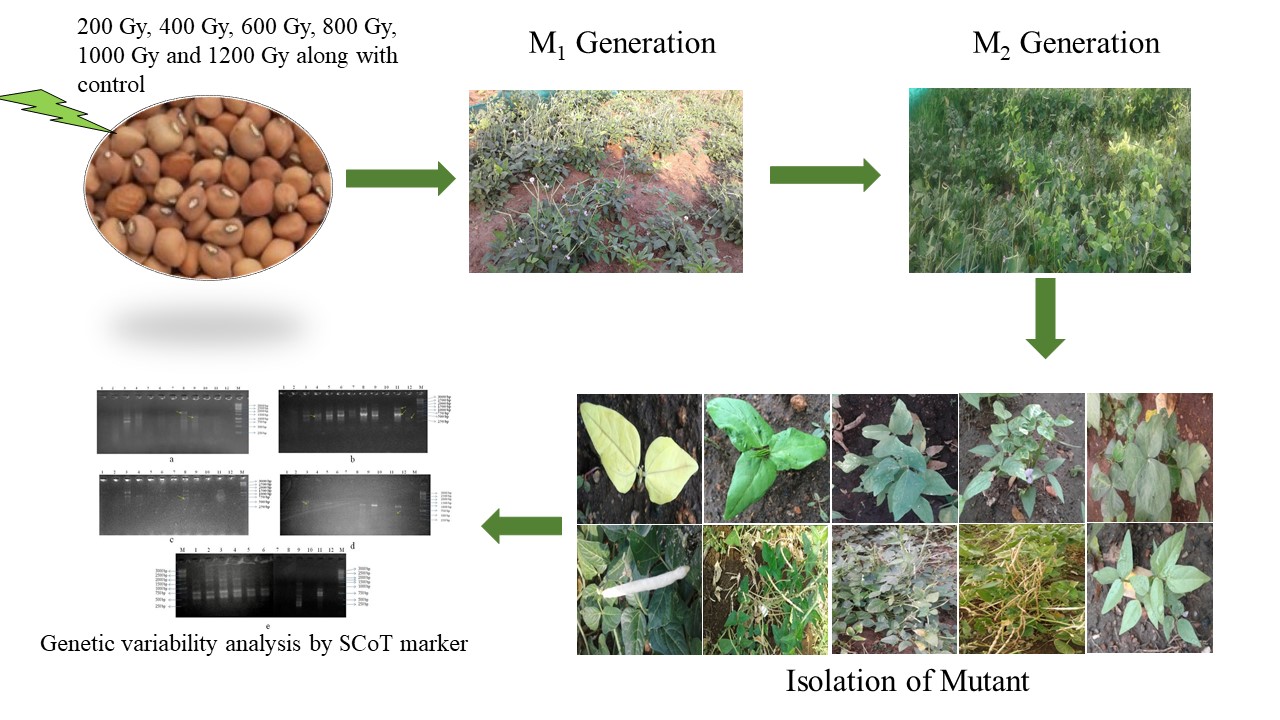
Mutagenesis is a well-known technique for introducing new variants into crop plants. In the present study, M2 populations were generated in the cowpea (Vigna unguiculata (L.) Walp.) variety CO7 using gamma irradiation. The M2 progeny were used to investigate the effectiveness of the gamma irradiation doses and examined for the agronomic traits. The genomic variation present in the mutants and their parents was analysed using five SCoT markers. Marker analysis revealed a total of 87 amplicons and among these, 20 amplicons showed polymorphism. The highest numbers of amplicons were observed at SCoT10 (39), while the lowest number of amplicons was produced by SCoT09 (07). The percentage of polymorphism ranged from 18.18% to 28.57%, with an average of 21.12%. Polymorphic information content (PIC) values ranged from 0.197 to 0.345. Analysis of Molecular Variation (AMOVA) showed 12% and 88% between the genotypes and within the genotypes respectively. The constructions of 4 clusters were identified through Unweighted Pair Group Method with Arithmetic Mean (UPGMA) dendrogram tree based on the genetic distance deduced from SCoT marker analysis. Analysis of the genetic relatedness between parent and mutants through Principal Coordinate Analysis (PCoA) revealed two main groups. The present study concludes that the genetic variability induced by gamma irradiation and inherited in the next generations. This research investigation supports that gamma irradiation alters the growth and yield traits, which is helpful for generating the cowpea improvement.
Downloads
References
Addo-Quaye AA, Darkwa A, Ampiah MKP. Performance of three cowpea ecological zones of the central region of Ghana: grain yield and its components. J Agri Bio Sci. 2011;6(2):34-42.
Agyeman K, Berchie JN, Osei-Bonsu I, Tetteh Nartey E, Fordjour JK. Growth and yield performance of improved cowpea (Vigna unguiculata L.) varieties in Ghana. Agri Sci. 2014;2(4):44-52.https://doi.org/10.12735/as.v2i4p44
Togola A, Boukar O, Belko N. Host plant resistance to insect pests of cowpea (Vigna unguiculata (L.) Walp.): achievements and future prospects. Euphytica. 2017;213-39.https://doi.org/10.1007/s10681-017-2030-1
da Silva AC, da Costa Santos D, Teixeira Junior DL, da Silva PB, dos Santos, RC, Siviero A. 2018. Cowpea: A strategic legume species for food security and health. Intech Open. 48-57: http://dx.doi.org/10.5772/intechopen.79006.
Chathuni J, Rizliya V, Afka D, Ruksheela B, Barana CJ, Srinivas N, Ruvini L. Cowpea: An overview on its nutritional facts and health benefits. Review. J Sci Food Agri. 2018;13:4793-806. https://doi.org/10.1002/jsfa.9074.
Boahen SK, Savala CEN, Chikoye D, Abaidoo R. Growth and yield responses of cowpea to inoculation and phosphorus fertilization in different environments. Front Plant Sci. 2017;8:646. https://doi.org/10.3389/fpls.2017.00646.
Vanmathi S, Arulbalachandran D, Soundarya V. Effects of gamma radiation on quantitative traits and genetic variation of three successive generations of cowpea (Vigna unguiculata (L.) Walp.). Plant Sci Today; 2021;8(3):578-89. https://doi.org/10.14719/pst.2021.8.3.1054
Arulbalachandran D, Mullainathan L. Chlorophyll and morphological mutants of blackgram (Vigna mungo (L.) Hepper) derived by gamma rays and EMS. J Phyto. 2009; 1(4):236-41.
Gharti DB, Darai R, Subedi S, Sarker A, Kumar S. Grain legumes in Nepal: Present Scenario and Future Prospects. World J Agric Res. 2014;2(5):216-22. https://doi.org/10.12691/wjar-2-5-3
Jiang SY, Ramachandran S. Natural and artificial mutants as valuable resources for functional genomics and molecular breeding. Int J Biol Sci. 2010;6:228-51. https://doi.org/10.7150/ijbs.6.228
Shah TM, Mirza JI, Haq MA, Atta BM. Induced genetic variability in chickpea (Cicer arietinum L.). II. Comparative mutagenic effectiveness and efficiency of physical and chemical mutagens. Pak J Bot. 2008;40(2):605-13.
International Atomic Energy Agency/ Mutant variety data base https://mvd.iaea.org/17th April 2020.
Garcia V, Bres C, Just D, Fernandez L, Tai FW, Mauxion J et al. Rapid identification of causal mutations in tomato EMS populations via mapping-by-sequencing, Nat Protoc. 2016;11:2401-18.https://doi.org/10.1038/nprot.2016.143
Pawar K, Yadav S, Arman M, Singh A. Assessment of divergence in soybean (Glycine max (L.) Merrill) germplasm for yield attributing traits. IJSR-Int J Sci Res. 2013;2:1-2.
Powell W, Morgante M, Andre C, Hanafey M, Vogel J, Tingey S, Rafalski A. The comparison of RFLP, RAPD, AFLP and SSR (microsatellite) markers for germplasm analysis. Mol Breed. 1996;2:225-38. https://doi.org/10.1007/BF00564200
Goncalves LS, Rodrigues R, do Amaral.Junior AT, Karasawa M. Heirloom tomato gene bank: assessing genetic divergence based on morphological, agronomic and molecular data using a Ward-modified location model. Genet Mol Res. 2009;8:364-74. https://doi.org/10.4238/vol8-1gmr549
Brown SM, Hopkins MS, Mitchell SE, Senior ML, Wang TY, Duncan RR et al. Multiple methods for the identification of polymorphic simple sequence repeats (SSRs) in sorghum (Sorghum bicolor (L.) Moench). Theor Appl Geneti. 1996;93:190-98. https://doi.org/10.1007/BF00225745.
Asare AT, Gowda BS, Galyuon IKA, Aboagye LM, Takrama JF, Timko MP. Assessment of the genetic diversity in cowpea (Vigna unguiculata (L.) Walp.) germplasm from Ghana using simple sequence repeat markers. Pl Gen Res. 2010;8(2):142–50. https://doi.org/10.1017/S1479262110000092
Ghalmi N, Malice M, Jacquemin JM, Ounane SM, Mekliche L, Baudoin JP. Morphological and molecular diversity within Algerian cowpea (Vigna unguiculata (L.) Walp.) landraces. Gen Res Crop Evo. 2010;57:371–86. https://doi.org/10.1007/s10722-009-9476-5
Malviya N, Yadav D, Sarangi BK, Yadav MK. Analysis of genetic diversity in cowpea (Vigna unguiculata (L.) Walp.) cultivars with random amplified polymorphic DNA markers. Pl Syst Evo. 2012;29:523-26. https://doi.org/10.1007/s00606-011-0545-9
Igwe DO, Afiukwa CA, Ubi BE, Ogbu KI, Ojuederie OB, Ude GN. Assessment of genetic diversity in Vigna unguiculata (L.) Walp. accessions using inter-simple sequence repeat (ISSR) and start codon targeted (SCoT) polymorphic markers. BMC Genet. 2017;18(1):1-13. https://doi.org/10.1186/s12863-017-0567-6
Ezzat A, Adly M, El-Fiki A. Morphological, agronomical and molecular characterization in irradiated Cowpea (Vigna unguiculata (L.) Walp.) and detection by start codon target markers. J Radiat Res Appl Sci. 2019;12(1):403-12. https://doi.org/10.1080/16878507.2019.1686578
Collard CY, Mackill DJ. Start Codon Targeted (SCoT) Polymorphism: A simple, novel DNA marker technique for generating gene-targeted markers in plants. Bertrand Plant Breeding, Genetics and Biotechnology Division, International Rice Research Institute (IRRI), Metro Manila, Philippines. 2009;27:86-93. https://doi.org/10.1007/s11105-008-0060-5
Joshi CP, Zhou H, Huang X, Chiang VL. Context sequences of translation initiation codon in plants. Plant Mol Biol. 1997;35:993-1001. https://doi.org/10.1023/A:1005816823636
Sawant SV, Singh PK, Gupta SK, Madnala R, Tuli R. Conserved nucleotide sequences in highly expressed genes in plants. J Genet. 1999;78:123-31. https://doi.org/10.1007/BF02924562
Rajesh MK, Sabana AA, Rachana KE, Rahman S, Jerard BA, Karun A. Genetic relationship and diversity among coconut (Cocos nucifera L.) accessions revealed through SCoT analysis. 3 Biotech. 2015;5:999-1006. https://doi.org/10.1007/s13205-015-0304-7
Nei M, Li WH. Mathematical model for studying genetic variation in terms of restriction endonucleases. In: Proceedings of the National Academy Sciences.1979;76:5269-73.https://doi.org/10.1073/pnas.76.10.5269
Peakall R, Smouse PE. GENALEX 6: genetic analysis in excel. Population genetic software for teaching and research. Molecular Ecology Notes. 2006;6:288-95. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Yasmin K, Arulbalachandran D. Effect of gamma rays on morphological and quantitative traits of black gram (Vigna mungo (L.) Hepper) in M1 generation. Inter J Cur Tre Res. 2016;4(2):5-12.
Yasmin K, Arulbalachandran D, Dilipan E, Vanmathi S. Characterization of 60CO ?-ray induced pod trait of black gram-A promising yield mutants. Inter J Radiat Biol. 2020;96(7):929-36. https://doi.org/10.1080/09553002.2020.1748738
Jegadeesan S, Reddy SK. Radiation induced mutations in genetic enhancement and development of new varieties in black gram (Vigna mungo (L.) Hepper). FAO/IAEA International Symposium on Plant Mutation Breeding and Biotechnology, Vienna, Australia. 2018. (No. IAEA-CN--263).
Goyal S, Wani MR, Khan S. Comparative mutagenic analysis of gamma rays, EMS and their combination treatments in black gram (Vigna mungo (L.) Hepper). Thai J Agric Sci. 2019;52(1):20-33.
Gupta PK, Rustgi S. Molecular markers from the transcribed/expressed region of the genome in higher plants. Funct Integr Genomics. 2004;4:139-62. https://doi.org/10.1007/s10142-004-0107-0
Varshney RK, Graner A, Sorrells ME. Genic microsatellite markers in plants: features and applications. Trends Biotechnol. 2005;23:48-55. https://doi.org/10.1016/j.tibtech.2004.11.005
Agarwal M, Shrivastava N, Padh H. Advances in molecular marker techniques and their applications in plant sciences. Plant Cell Rep. 2008;27:617-31.https://doi.org/10.1007/s00299-008-0507-z
Madhumati B. Potential and application of molecular markers techniques for plant genome analysis. Int J Pure Appl Biosci. 2014;2(1):169-88.
Arulbalachandran D, Mullainathan L, Velu S. Genetic variation in quantitative traits of black gram (Vigna mungo (L.) Hepper) induced by gamma rays treatment in M3generation. J Phytol. 2009;1(5):308-12.
Mudibu J, Nkongolo KK, Mehes-Smith M, Kalonji-Mbuyi A. Genetic analysis of a soybean genetic pool using ISSR marker: Effect of gamma radiation. Int J Plant Breed. 2011;5:235-45. https://doi.org/10.3923/ijpbg.2011.235.245
Souframanien J, Pawar SE, Rucha AG. Genetic variation in gamma ray induced mutants in black gram as revealed by RAPD and ISSR markers. Indian J Genet Plant Breed. 2002;62(4):291-95.
Dhakshanamoorthy D, Selvaraj R, Chidambaramb A. Utility of RAPD marker for genetic diversity analysis in gamma rays and ethyl methane sulphonate (EMS)-treated Jatropha curcas plants. C R Biol. 2014;338:75-82.https://doi.org/10.1016/j.crvi.2014.12.002
Ginchner T, Zindar I, Szakova J. Evaluation of DNA damage and mutagenicity induced by lead in tobacco plants. Mutat Research. 2008;652:186-90. https://doi.org/10.1016/j.mrgentox.2008.02.009
Atienzar FA,Jha AN. The random amplified polymorphic DNA (RAPD) assay and related techniques applied to genotoxicity and carcinogenesis studies: A critical review. Mut Res. 2006;613:76-102.https://doi.org/10.1016/j.mrrev.2006.06.001
Nagy S, Poczai P, Cernak I, Gorji AM, Hegedus G, Taller J. PICcalc: An online program to calculate polymorphic information content for molecular genetic studies. Biochem Genet. 2012;50:670-72.https://doi.org/10.1007/s10528-012-9509-1
Bibi S, Khan IA, Bughio H, Odhano IA, Asad MA,Khatri A. Genetic differentiation of rice mutants based in morphological traits and molecular marker (RAPD). Pak J Bot. 2009;41:737-43.
Kolade OA, Olowolafe MO, Fawole I. Characterization of mutant cowpea (Vigna unguiculata (L.) Walp.) lines using random amplified polymorphic DNAs (RAPDs) and amplified fragment length polymorphism (AFLP) markers. Afr J Biotechnol. 2016;15:2530-37.https://doi.org/10.5897/AJB2015.14539
Sianipar NF, Laurent D, Purnamaningsih R, Darwati I. Genetic Variation of the First Generation of rodent tuber (Typhonium flagelliforme Lodd.) mutants based on RAPD molecular markers. HAYATI J Biosci. 2015;22(2):98-104. https://doi.org/10.4308/hjb.22.2.98.
Downloads
Published
Versions
- 01-07-2022 (2)
- 30-05-2022 (1)
How to Cite
Issue
Section
License
Copyright (c) 2022 Vanmathi Sankar, Arulbalachandran Dhanarajan; Selvakumar Gurunathan, Dilipan Elangovan

This work is licensed under a Creative Commons Attribution 4.0 International License.
Copyright and Licence details of published articles
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
Open Access Policy
Plant Science Today is an open access journal. There is no registration required to read any article. All published articles are distributed under the terms of the Creative Commons Attribution License (CC Attribution 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited (https://creativecommons.org/licenses/by/4.0/). Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).








